|
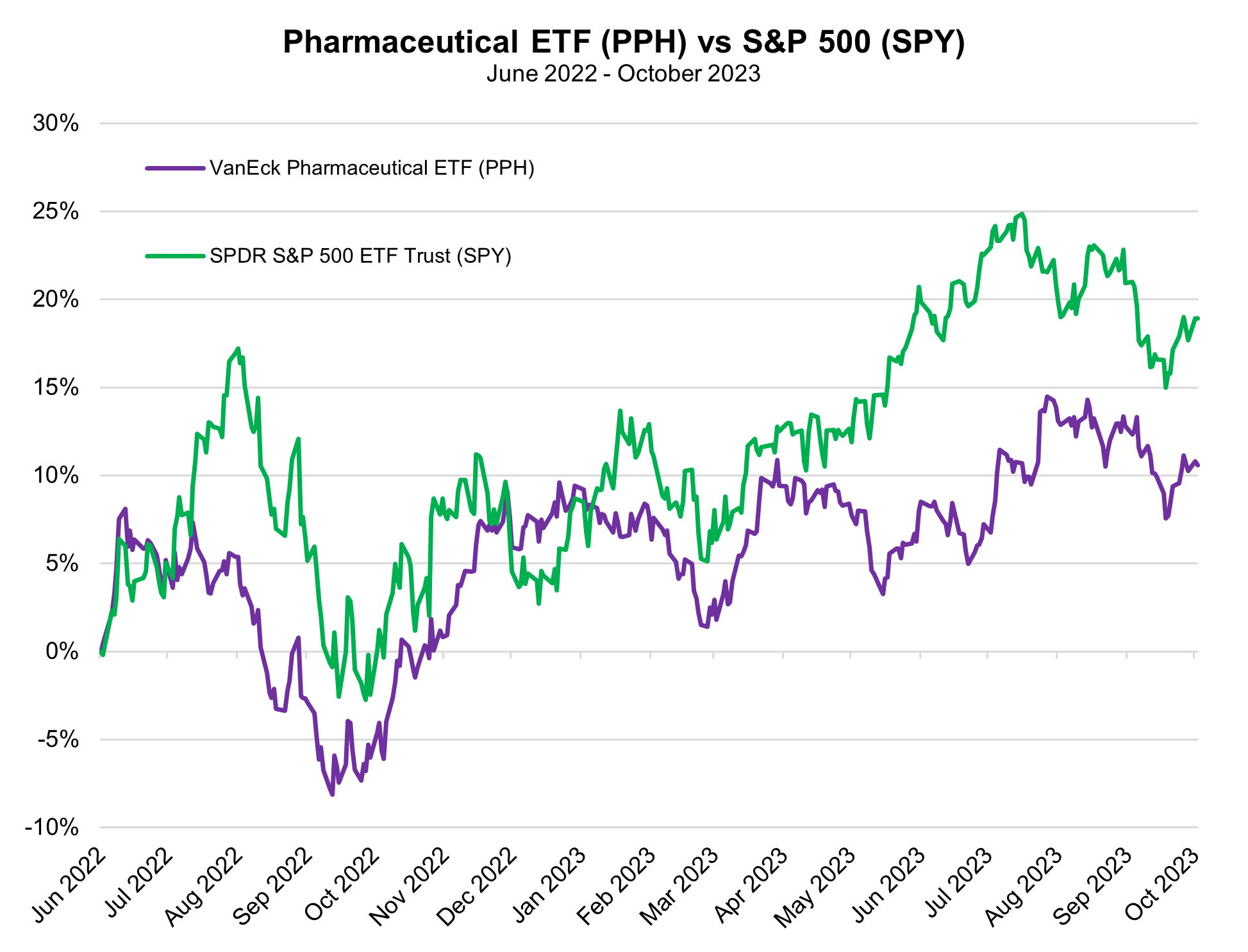
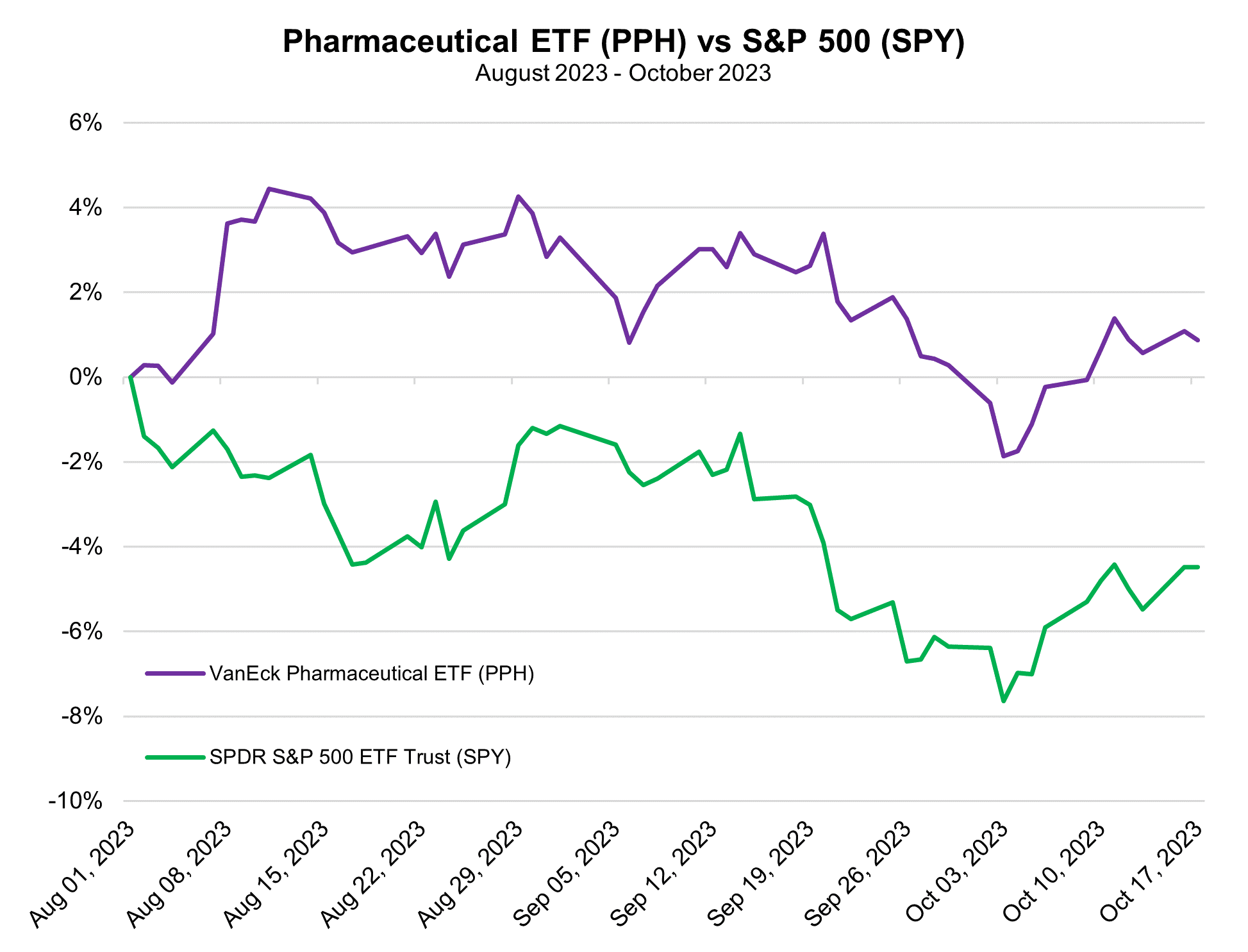
Manufacturers of US-approved COVID-19 vaccines, like Pfizer and Moderna, have gone through a boom and bust of pandemic-related product sales, but may finally be turning the corner on the COVID era, looking toward promising projects in their pipelines to revive their financial performance.
Pfizer, for instance, announced late last Friday that the company would be slashing its 2023 revenue projection by $9 billion due to declining demand for its COVID vaccine shots, Comirnaty, and the Paxlovid treatment. These two drugs were the primary products that pushed Pfizer to become the first company in the history of the biopharma industry to top $100 billion in sales back in 2022. A combined $57 billion in revenues were derived from Comirnaty and Paxlovid that year. CEO Albert Bourla said on Monday that just 17% of the US population to get updated COVID-19 vaccines during the current vaccination campaign, in-line with last year, but failing to retain most of the 81% of the population that received at least one dose of the COVID vaccines at some point in the past two-and-a-half years.
Pfizer also sees earnings per share plummeting to between $1.45 and $1.65, down from its previous projection of $3.25 to $3.45. While these are far from positive developments, the announcement cleared the air of uncertainty that was looming ahead of its upcoming third quarter earnings call on October 31, putting a figure on just how bad things may get for Pfizer’s projections before they start to improve. Pulling off the band-aid on COVID sales, as well implementing a $3.5 billion cost-cutting plan, actually boosted Pfizer shares on Monday, which helped to offset a small portion of the declines the company has faced in 2023. These have eaten away more than a third of the company’s market cap in the year-to-date period.
To rebuild itself and look ahead, Pfizer will need to bolster their pipelines with new internal developments, as well as outside acquisitions. As MRP has previously noted, Pfizer executives have been among a consortium of biopharma heads that have voiced their desire to increase dealmaking with outside companies. Pfizer has set a goal of adding $25 billion in revenue by 2030 from business-development moves including acquisitions. Those could help the company offset an estimated drop of roughly $17 billion in sales from upcoming patent expirations. The company was the purchasing party in two of the top three largest pharma M&A deal disclosures of 2022, each worth $11.6 and $5.4 billion. Further, they are now in the process of closing its acquisition of massive biotech firm Seagen Inc. in biopharma’s largest deal in almost four years’ time. Pfizer’s offer of $229 per share in cash, a 33% premium on Seagen’s share price at the close preceding the deal becoming public, pushed total value of the deal to $43 billion.
The prospects for this deal were previously thrown into uncertainty by FTC legal action that put a hold on Amgen’s $27.8 billion purchase of Horizon Therapeutics, but a settlement in that case appeared to defang the threat of the commission taking similar action on the Pfizer-Seagen deal. Bloomberg has noted that Pfizer sees sales of Seagen’s four FDA-approved oncology products exceeding $10 billion, about $2 billion more than analysts’ estimates. Pfizer’s Velsipity, a drug to treat ulcerative colitis, was approved by the FDA just last week, a testament to Pfizer’s M&A strategy bearing fruit. Velsipity was a key asset that Pfizer picked up in its deal for Arena Pharmaceuticals, which closed last year. The risk-adjusted sales of Pfizer’s drug are estimated to bring in $2.2 billion of revenue by 2030. Future growth opportunities for the company include the launch of a new vaccine for respiratory illness RSV and an experimental obesity drug with data expected later this year.
Moderna, another biopharma that benefited big time from COVID vaccine sales, has seen an even larger sell-off of their shares than Pfizer. The company’s market cap has been cut in half throughout 2023 with vaccine sales falling. In the wake of Pfizer’s announcement that it would be cutting its COVID-related sales, Moderna has said it remains comfortable with its prediction of $6 billion to $8 billion in vax sales. That range assumes a broad reach of 50 million to 100 million doses will be distributed in the fall vaccination campaign. Though the vaccine maker is still very reliant on this optimistic outlook for COVID, it has worked throughout 2023 to develop new breakthroughs on vaccines targeting cancer, heart disease, and other conditions.
Moderna, founded in 2010, made its first acquisition early this year, expending $85 million to purchase OriCiro Genomics – meant to speed development of Moderna’s pipeline by offering better tools to synthesize and amplify plasmid DNA. Last month, Moderna struck a partnership with Immatics, offering up the latter’s IMA203 PRAME TCR-T for combination with Moderna’s investigational PRAME mRNA cancer vaccine. Moderna is paying $120 million up front to Immatics and then up to $1.7 billion in development, regulatory and commercial milestones down the line. That builds on a similar partnership Moderna has forged with Merck, combining Moderna’s mRNA-based skin cancer vaccine with the immunotherapy Keytruda. Late-stage studies on this treatment began in July, following a mid-stage study that found the vaccine combination cut the risk of recurrence or death by 44% in patients with melanoma, the most deadly form of skin cancer. Merck paid $250 million for joint rights to the personalized cancer vaccine now in development by the two companies in 2022.
|



Leave a Reply